I’m getting closer to the point where amino acids make sense to me. Right now, I can look at chemical formulas and pick out which chemicals I think are amino acids and which ones are definitely not. That’s progress.
But I still have a ways to go. So far here on the blog, I’ve introduced you to amino groups and carboxyl groups. By definition, amino acids must include at least one of each of these functional groups. So let’s stick a carboxyl group to an amino group and see what happens.
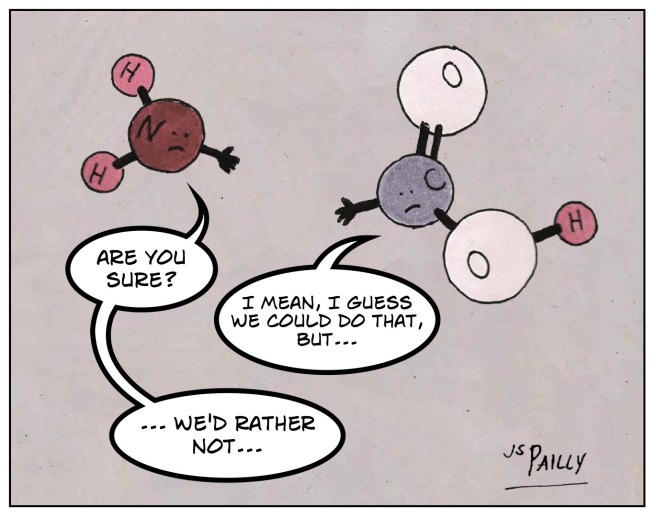
Oh, come on, guys! You each have one available bonding site. What’s the problem?

This is a chemical called carbamic acid. The “carb” part of the name comes from “carboxyl,” and the “am” part is for “amino.” Although this is not considered an amino acid, it has a pair of functional groups that should still be useful for biological processes. They should be, except…
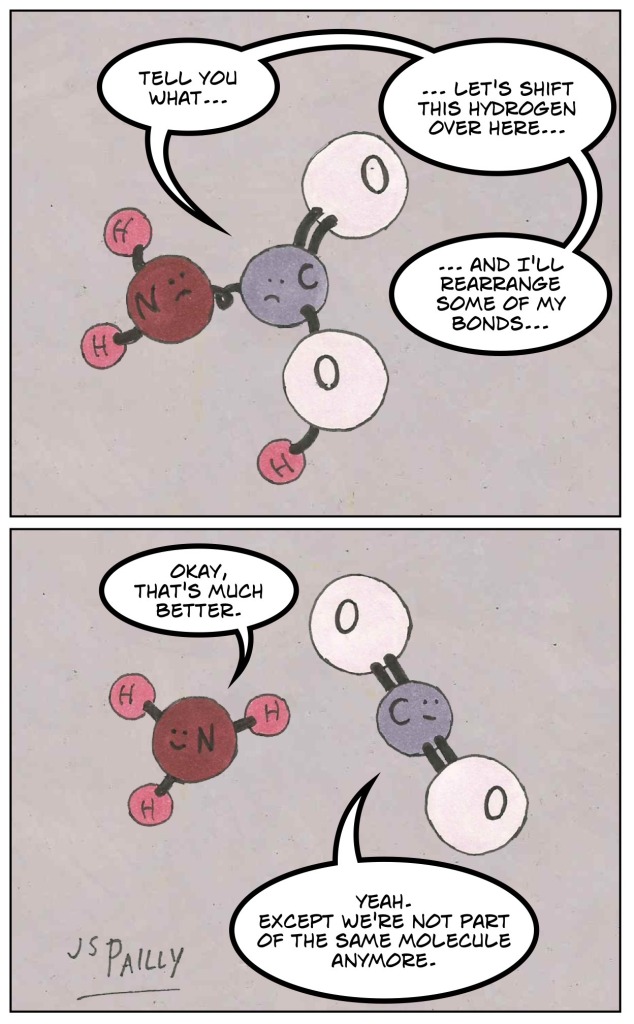
So apparently carbamic acid has a tendency to reshuffle its constituent atoms, trying to find a more comfortable and stable arrangement. In the process, the molecule just falls apart, leaving us with ammonia and carbon dioxide.
There’s something that always sort of bugged me about chemistry. With over 90 different naturally occurring elements and so many different kinds of chemical bonds that can form between them, there should be a virtually infinite number of molecules. The larger and more complicated molecules are, the more varieties should be possible.
So how come certain molecules, molecules which seem to make sense on paper, are so rare in nature or don’t seem to exist at all? Perhaps the example of carbamic acid provides a clue.
* * *
Today’s post is part of a special series here on Planet Pailly called Molecular Mondays. Every other Monday, I struggle valiantly to understand and explain some concept in the field of chemistry. Please note: I suck at chemistry, but I’m trying to learn. If I made a mistake, please, please, please let me know so I can get better.
This reminds of something Sean Carroll once noted, that we don’t know enough yet to take the periodic table and calculate the universe, at least not without fudging on many of the steps.
LikeLiked by 1 person
I hadn’t heard that before, but it certainly seems true.
LikeLiked by 1 person